Division of Pulmonary, Critical Care, Sleep and Allergy Research
The Division of Pulmonary, Critical Care, Sleep and Allergy members are highly engaged in advancing the science of practice of medicine with diverse areas of expertise including vascular biology, immunology, clinical outcomes and implementation research spread across pulmonary, critical care, sleep and allergy specialties. Our faculty are nationally and internationally recognized as leaders in their respective fields. The specific programs in the Division concentrate in Pulmonary Hypertension, ARDS and Lung Injury, Lung cancer, COPD and Asthma, Sleep Disorders and Nanomedicine and Drug Discovery. We have a very active portfolio of research projects, funded by the National Institutes of Health, VA system, PCORI, AHA, RHA, and various other private foundations or pharmaceutical companies.
In FY2020, the total number of active external grants increased to 56. Our entire grant profile totals more than $13 million annually (direct and indirect funding). The highly productive members of the Division published 94 original scholarly works in FY 2020, including multiple publications in high impact journals. This active scholarship produces many collaborative pursuits within the Departments of Medicine, Pharmacology the School of Public Health, and others here at UIC which have generated national and international recognition. As noted above, we are particularly proud of our success in training and nurturing the next generation of pulmonary-focused scientists, with our Division earning multiple NIH K level Career Development awards in the past several years, which pursued important research questions relevant to a diverse array of clinical problems—asthma, ARDS, pulmonary hypertension, and COPD.
Research Topics Heading link
-
Research Topics
ARDS / Lung Injury
Patrick Belvitch
Steven Dudek
Jeffrey Jacobson
Viswanathan Natarajan
Sunit SinglaCOPD / Asthma
Steven Dudek
Jeffrey Jacobson
Jerry Krishnan
Stephanie LaBedz
Israel Rubinstein
Viswanathan Natarajan
Andrea A. PappalardoLung Cancer
Kevin Kovitz
Mary Pasquinelli
Viswanathan Natarajan
Israel RubinsteinNanomedicine Drug Discovery Entrepreneurial Activities
Steven Dudek
Israel Rubinstein
Sunit SinglaPulmonary Hypertension
Dustin Fraidenburg
Irena Levitan
Viswanathan NatarajanSleep Disorders
Ikuyo Imayama
Vascular Biology
Steven Dudek
Dustin Fraidenburg
Jeffrey Jacobson
Irena Levitan
Viswanathan Natarajan
Sunit SinglaSarcoidosis / ILD
Steven Dudek
Jeffrey Jacobson
Israel Rubinstein
Viswanathan NatarajanInterventional pulmonary
Kevin Haas
Chris Kapp
Kevin KovitzCOVID-19, including Post-acute Sequelae of SARS-CoV-2
Israel Rubinstein
Jerry Krishnan
Sunit SinglaBronchopulmonary dysplasiato
Viswanathan Natarajan
Pulmonary Toxicities of E-cigarette Aerosols
Sunit Singla
-
Research
Basic-Translational Research
Patrick Belvitch, MD
Steven Dudek, MD, ATSF
Dustin Fraidenburg, MD
Jeffrey Jacobson, MD
Irena Levitan, PhD
Viswanathan Natarajan, PhD
Gye Young Park, MD
Sunit Singla, MDClinical-Outcomes Research
Kevin Haas, MD
Min Joo, MD
Kevin Kovitz, MD, MBA
Jerry A. Krishnan, MD, PhD
Marisol Munoz, MSN, FNP-BC
Mary Pasquinelli, DNP, FNP-BC
Bharati Prasad, MD
Israel Rubinstein, MDHealth Services Research
Research Awards/Grants Heading link
-
New Grants
Stephanie LaBedz, MD
CCTS KL2 CATS Scholar
Project title: Improving Inhaler Adherence in Underserved Patients with Chronic Obstructive Pulmonary Disease
New important publication from the group of Dr. Gye Young Park
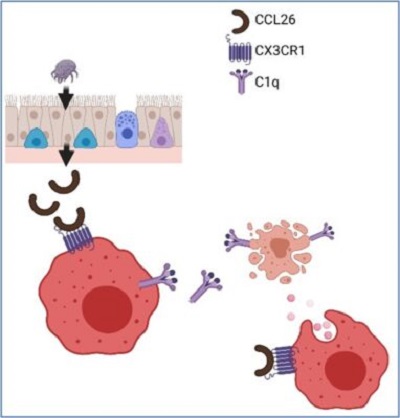
CX3CR1+ Macrophage Facilitates the Resolution of Allergic Lung Inflammation via Interacting CCL26
Am. J. Respir. Crit. Care Med. 2023The resolution of inflammation not only ceases the inflammatory processes but also requires active processes to restore tissue homeostasis. This is especially true in the cases of inflammation associated with neutrophils and eosinophils because these granulocytes are short-lived cells and undergo apoptotic cell death rapidly. Restoring normal hemostasis won’t be easy after the insults without proper mechanisms to clear dead bodies of tissue-infiltrated granulocytes. The failure of this clearing process could prolong inflammation and lead to chronic inflammatory diseases in humans. Tissue-resident macrophages have been known for this job in neutrophilic conditions because of their high phagocytic capabilities. However, the identity of tissue macrophages for clearing tissue-recruited eosinophils was unknown until we published this paper. Our group, including our lab and collaborating investigators, has been working on searching for the lung macrophages responsible for removing eosinophils in eosinophilic-allergic lung inflammation. Using the human samples from the IRB-approved translational protocol and relevant animal models, we found that the CX3CR1+ macrophage, a subset of lung macrophages, is the one responsible for removing eosinophils in the allergic lungs. Furthermore, applying sophisticated analytic tools such as single cell-RNA sequencing and the SPR method, we discovered the mechanism of how CX3CR1+ macrophages are activated, as shown in the figure. The research highlights the diversity of lung-resident macrophages in terms of their functions and phenotypes. Finally, the discovery could lead to new therapeutic approaches for various inflammatory lung diseases.
Andrea Pappalardo
 Andrea Pappalardo, MD
Andrea Pappalardo, MDAndrea A. Pappalardo, MD is an Assistant Professor of Medicine and Pediatrics in the Division of Pulmonary, Critical Care, Sleep and Allergy at the University of Illinois Chicago–UI Health. She has been practicing clinical allergy immunology across the lifespan in a variety of settings including: urban, suburban and community practice for over ten years. She is the Medical Director of the Mobile Care Chicago Asthma and Allergy Program, and the Medical Director of CHECK, a care coordination program for Medicaid recipients in Illinois. Dr. Pappalardo is now shifting her career focus to improving healthcare access through implementation-science based healthcare innovation. Her K12 project is to assess school-based asthma management practices in Chicago Public Schools and pilot a stock inhalers program in select Chicago schools with demonstrated high asthma burden. Health equity is central to Dr. Pappalardo’s work as she focuses on health-related policy implementation.
-
New Awards (awarded in 2022)
Career K awards
AHRQ, K12, Dr. Andrea Pappalardo, MD
School-Based Asthma Management in Chicago Public Schools: An Implementation Science-Based Approach to Coordinated Asthma Care, 2022-2024CCTS KL2 CATS Scholar Notice of Award, PI: Christian Ascoli, MD
Mechanisms and Clinical Relevance of Lymphopenia in Sarcoidosis.\, 2022-2023AHA Career Development Award PI: Eleftheria Letsiou, PhD
Role of extracellular vesicles in alveolar-capillary crosstalk in acute lung injury, 2022-2025Clinical-Outcomes Research
Insmed Incorporated, PI: Dustin Fraidenburg, MD
A Randomized Double Blind Placebo Controlled Study of Treprostinil Palmitil Inhalation Powder in Participants with Pulmonary Arterial Hypertension, 2021-2023Lung Biotechnology PBC, PI: Dustin Fraidenburg, MD
An Open-Label Extension Study of Inhaled Treprostinil in Patients with Pulmonary Hypertension due to Chronic Obstructive Pulmonary Disease (PH-COPD), 2019-2024Basic-Translational Research Grants
R01 NHLBI PI: Asrar Malik, MD and Steven Dudek, MD Core Lead
Macrophage Plasticity In Inflammatory Lung Injury – Patient Sampling and Genomics Core Project, 2021-2026UI Health Equity Pilot Project (HEPP), PI: Christopher Kapp, MD
Improving Access to Appropriate Diagnostics and Treatment in a High-Risk Population, 2022-2023 -
Additional Active Awards