Veiga-Lopez Lab Research
Placental programming of adult disease Heading link
Despite its central role during pregnancy, the placenta is often overlooked in the Developmental Origins of Health and Disease (DoHAD) field and toxicological studies. To understand how endocrine disrupting chemicals can affect placental function contributing to this higher incidence in pregnancy disorders and the role the placenta plays in fetal programming, our laboratory is investigating how emerging bisphenolic endocrine disrupting chemical may induce placental dysfunction and ultimately affect fetal development. Our work has shown that bisphenol S, an emerging bisphenol, reduces the endocrine capacity of the placenta (Gingrich et al., Archives of Toxicology, 2018) and that it can cross the placental barrier reaching fetal circulation (Gingrich et al., Chemosphere, 2019). Our pregnancy PBPK model shows that BPS is more likely to accumulate in the fetal compartment compared to BPA (Gingrich et al., Environment International 2021). More recently, we have demonstrated that BPS interferes with human placental cell fusion (Ticiani et al., Environmental Health Perspectives, 2021) and placental cell invasion (Ticiani et al., IJMS 2022) through the epidermal growth factor receptor pathway.
Check out our latest review on the effects of endocrine disrupting chemicals on placental function (Placenta disrupted: endocrine disrupting chemicals and pregnancy. Trends in Endocrinology and Metabolism 2020).
We are continuing to explore the mechanisms by which other emerging endocrine disrupting chemicals can alter placental function. In these studies we use animal models, primary cultured cells, cell lines, and 3-dimensional microfluidic platforms (Pu et al. Lab on a Chip, 2021).
This work is currently funded by an NIEHS/NIH R01 award.
Bisphenol S reduces e-cadherin expression in the placenta (Arch Tox 2018) Heading link
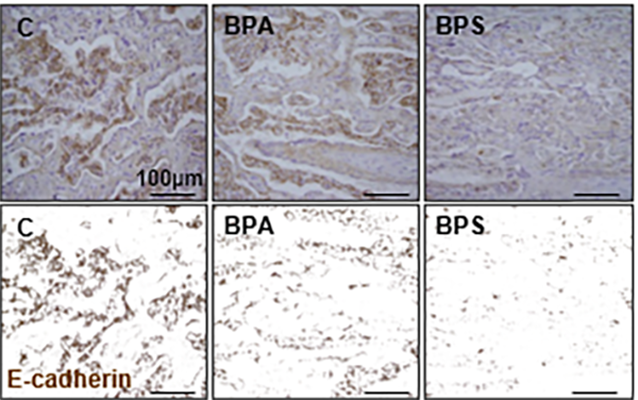
Bisphenol S reduces e-cadherin expression in the placenta (Arch Tox 2018)
3-D microfluidic system for placenta trophoblast cell invasion and cell-to-cell interaction studies under dynamic environment conditions (Lab on a Chip, 2021) Heading link
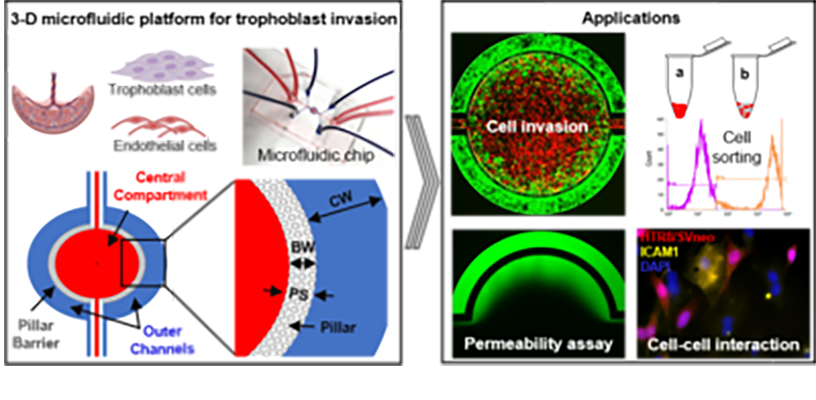
3-D microfluidic system for placenta trophoblast cell invasion and cell-to-cell interaction studies under dynamic environment conditions (Lab on a Chip, 2021)
BPS blocks EGF-mediated human cytotrophoblast (hCTB) cell fusion (Env. Health Perspect. 2021) Heading link
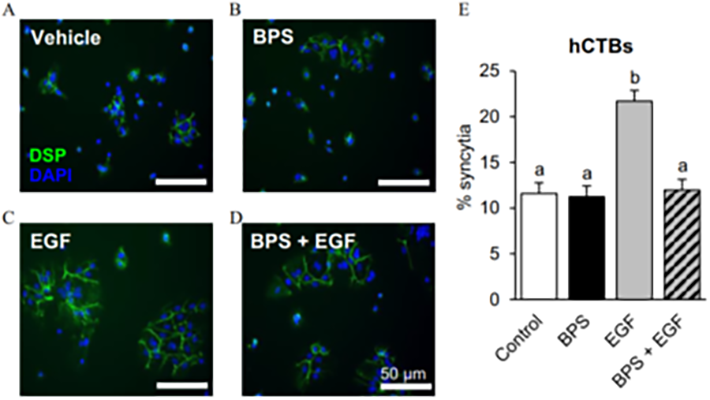
BPS blocks EGF-mediated human cytotrophoblast (hCTB) cell fusion (Env. Health Perspect. 2021)
Pregnancy toxicokinetic model for bisphenols (Gingrich et al., Environment International 2021). Heading link
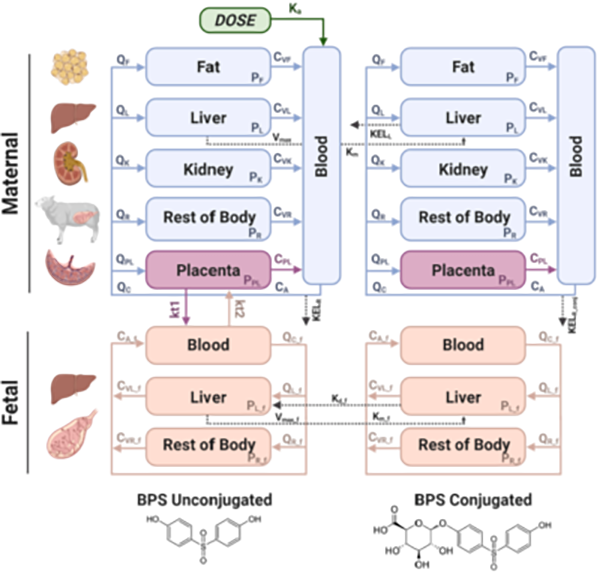
Pregnancy toxicokinetic model for bisphenols (Gingrich et al., Environment International 2021).
Fetal origins of metabolic disease Heading link
Fetal exposure to the endocrine disrupting chemical bisphenol A (BPA) causes insulin resistance in the adult (Veiga-Lopez et al., Am J Physiol – Endocrinol Metab 2016), a pre-stage of type 2 diabetes, which affects ~350 million people. In women, gestational BPA exposure affects the newborn’s weight in a sex-specific manner (Veiga-Lopez et al., J Clin Endocrinol Metab 2015). Animal studies suggest that weight increases are due to increase body fat. How BPA induces insulin resistance and weight gain/obesity remains unknown. Our research using in vivo and cell-based assays aims to understand the mechanistic basis by which BPA programs insulin resistance and obesity during fetal life. Our work has demonstrated for the first time that gestational BPA can increase the ability of fetal primary preadipocytes to differentiate into mature adipocytes and points to the endoplasmic reticulum stress as a potential modulator in this effect. (Pu et al., Endocrinology 2017). Our recent work has also focused on another insulin target tissue, the skeletal muscle. We have demonstrated sex- and bisphenol-specific effects on myocytes upon prenatal exposures (Jing et al., Toxicological Sciences 2020).
Check out our review on the effects of endocrine disrupting chemicals on adipogenesis (Obesogenic endocrine disrupting chemicals: identifying knowledge gaps. Trends in Endocrinology & Metabolism 2018).
This work was funded by an NIEHS/NIH K22 TIEHR award.
Gestational BPA leads to increased fetal adipogenic differentiation. Heading link
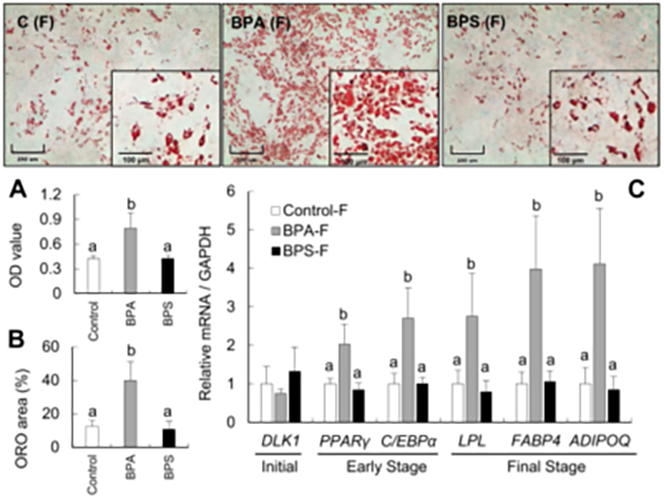
Gestational BPA leads to increased fetal adipogenic differentiation.
BPA and BPS alters myogenic differentiation during fetal life. Heading link
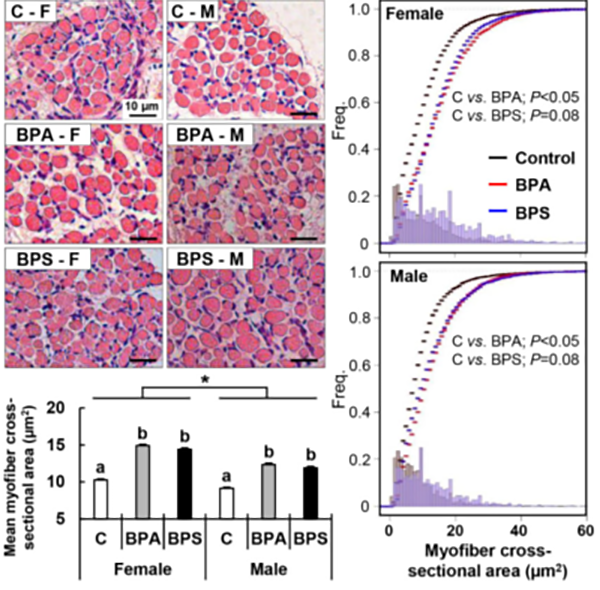
BPA and BPS alters myogenic differentiation during fetal life.
Improving small ruminants intensive production systems Heading link
As a veterinarian, I also have a substantial interest in applied research to improve sheep production systems, an outstanding priority for the Michigan Sheep Breeders Association. Given how maternal nutritional status and environment affect offspring health, my laboratory is partnering with Dr. Richard Ehrhardt (Michigan State University) and conducting studies focusing on the effects of plane of nutrition and its relationship with breeding season in an intensive sheep production system at the Sheep Teaching & Research Center. Some of our work relates to placental endocrine function (Roberts et al., Theriogenology 2017), effects of production practices on placental function (Rosales-Nieto et al., 2020) and how maternal nutrition affects placental function and offspring growth (Rosales-Nieto et al., 2021).
This work is currently funded by the Michigan Alliance for Animal Agriculture.
Dynamic change in pregnancy associated glycoproteins (PAG1 & PSBP) through pregnancy (Roberts et al., 2017) Heading link
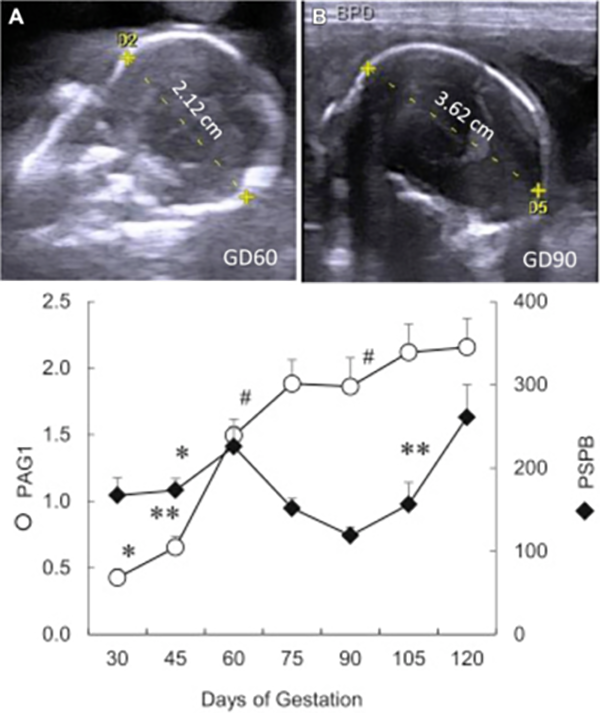
Dynamic change in pregnancy associated glycoproteins (PAG1 & PSBP) through pregnancy (Roberts et al., 2017)
Pregnancy associated glycoproteins reach baseline 10 weeks postpartum in the mother and less than10 days in the newborn (Roberts et al., 2017) Heading link
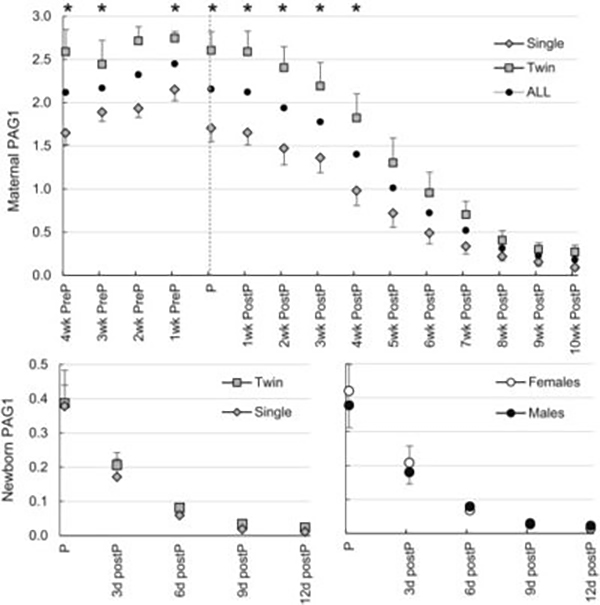
Pregnancy associated glycoproteins reach baseline 10 weeks postpartum in the mother and less than10 days in the newborn (Roberts et al., 2017)